Matter is everything that we see around us in our daily life. From the air we breathe to the water we drink, everything is made up of matter. In Class 9 Science, the chapter “Matter in Our Surroundings” helps students understand the basic nature, properties, and different states of matter in a simple way. This topic is very important because it forms the foundation for higher-level Chemistry concepts.
In this blog post, you will learn what matter is, how particles behave, different states of matter, and how matter changes from one state to another. The concepts are explained in easy language with real-life examples so that every student can understand quickly. These notes are perfect for revision, exams, and building a strong base in science.
Matter in Our Surroundings – Short Notes (Class 9)
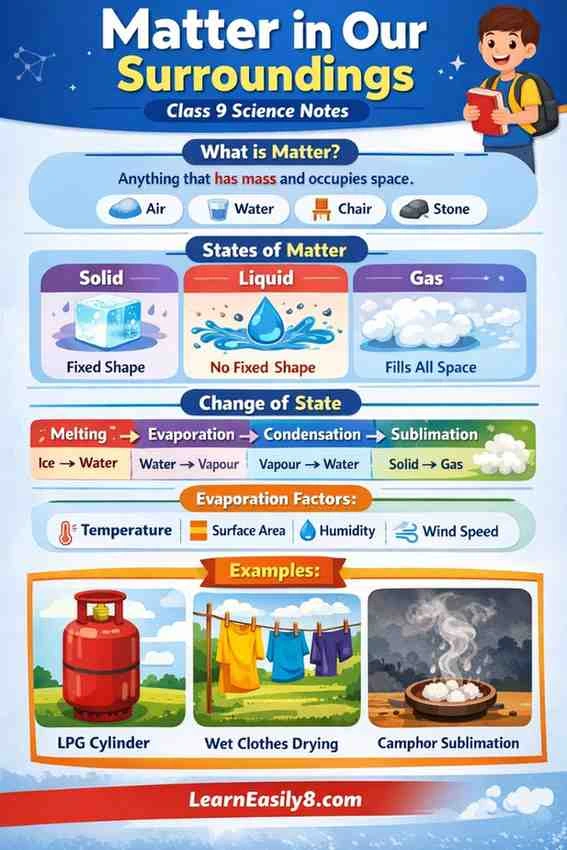
What is Matter?
Anything that has mass and occupies space is called matter.
Examples: Air, water, stone, milk, chair, etc.
Physical Nature of Matter
- Matter is made up of tiny particles
These particles are very small and cannot be seen with naked eyes.
Example: Sugar dissolves in water but still exists. - Particles of matter have spaces between them
Example: When salt is added to water, it disappears in the spaces. - Particles of matter are continuously moving
This movement is called kinetic energy.
Example: Smell of perfume spreads in a room. - Particles attract each other
There is a force of attraction between particles.
Example: It is hard to break a solid like iron due to strong attraction.
States of Matter
There are mainly three states of matter:
1. Solid
- Fixed shape and volume
- Particles are tightly packed
- Very strong force of attraction
Example: Ice, wood, stone
2. Liquid
- Fixed volume but no fixed shape
- Takes shape of container
- Particles are less tightly packed
Example: Water, milk, oil
3. Gas
- No fixed shape and no fixed volume
- Particles are far apart and move freely
Example: Air, oxygen, carbon dioxide
Change of State of Matter
Matter can change from one state to another by changing temperature or pressure.
Important Processes:
There are the following methods through which a matter can change into other state.
- Melting: Solid → Liquid
👉 Ice → Water - Freezing: Liquid → Solid
👉 Water → Ice - Evaporation: Liquid → Gas
👉 Water → Vapour - Condensation: Gas → Liquid
👉 Vapour → Water droplets - Sublimation: Solid → Gas (directly)
👉 Example: Camphor, dry ice
Effect of Temperature
- On heating, particles gain energy and move faster
- On cooling, particles lose energy and come closer
Example: Ice melts into water when heated
Effect of Pressure
- Increasing pressure can convert gases into liquids
Example: LPG cylinder contains gas in liquid form due to high pressure
Evaporation
Evaporation is a surface phenomenon where liquid changes into gas at any temperature.
Factors affecting evaporation:
- Temperature (higher → faster evaporation)
- Surface area (larger → faster)
- Humidity (less humidity → faster)
- Wind speed (higher → faster)
Example: Wet clothes dry faster in sunlight and wind
Latent Heat
The heat required to change the state of matter without changing temperature is called latent heat.
Example: Ice melts at 0°C but temperature does not increase until all ice melts.
Matter in Our Surroundings: Conclusion
Matter is present all around us in different forms. Its properties depend on how particles are arranged and how they move. Understanding matter helps us explain many daily life processes like melting, boiling, and evaporation. For more details contact Learneasily8.
Matter in Our Surroundings: FAQ
What is matter in simple words?
Matter is anything that has mass and occupies space.
What are the 3 states of matter?
Solid, Liquid, and Gas.
What is evaporation?
Evaporation is the process of converting liquid into gas at any temperature.
Give example of sublimation
Camphor changes directly into gas.

